The perianal, perineal, and perigenital regions of the dog represent an anatomical territory whose complexity is often underestimated in routine consultations. Yet the dermatoses occurring in these areas are common, polymorphic, and capable of generating significant morbidity. Intractable pruritus, chronic pain, ulcerative or nodular lesions: these are all clinical presentations that require a structured diagnostic approach. At the most recent ESVD congress, our colleague Elisa Maina from the University of Bern provided a comprehensive overview of these various dermatoses.
The multiplicity of anatomical structures concentrated in this area — anal sacs, hepatoid glands, keratinized squamous epithelium, and external genitalia — explains the nosological diversity of the conditions encountered. From simple allergic pruritus to neoplasia with high metastatic potential, including immune-mediated perianal fistula, each clinical entity requires precise recognition and appropriate therapeutic management. The perineal region, for its part, is distinguished by its permanent exposure to fecal matter, urine, and exogenous irritants, making it a zone of particular dermatological vulnerability. The perigenital region, which includes the external genitalia of both sexes, has its own clinical specificities, ranging from vulvar intertrigo in the female to scrotal dermatitis in the male.
Anatomical Foundations: An Essential Clinical Prerequisite
Architecture of the Perianal Region
The perianal region is structurally more complex than its external appearance alone suggests. The anus is subdivided into three functionally and histologically distinct zones. The columnar zone, the most proximal, is directly connected to the rectal mucosa. The intermediate zone represents a transitional area. Finally, the cutaneous zone, consisting of keratinized squamous epithelium, is of primary clinical importance: it is here that the hepatoid glands — also referred to as circumanal or perianal glands — are located, along with the openings of the excretory ducts of the anal sacs.
This anatomical distinction is not trivial. Many conditions show a selective tropism for one or another of these sub-zones, which directly influences their clinical expression and differential diagnosis. Confusing involvement of the cutaneous zone with strictly mucosal or rectal pathology constitutes a diagnostic error with potentially detrimental therapeutic consequences. The perianal cutaneous zone, by virtue of the nature of its keratinized epithelium and the density of glandular structures it contains, represents the preferred site for hepatoid gland neoplasias and chronic inflammatory processes such as perianal fistula. Its precise clinical recognition is therefore an indispensable prerequisite for any serious diagnostic approach in this region.
The Anal Sacs: Structure, Contents, and Physiology
The anal sacs, also called perianal sacs, are two bilateral pouches nestled in the subcutaneous tissue, whose excretory ducts open precisely into the cutaneous portion of the anus. Their wall is composed of two glandular types: sebaceous glands, distributed mainly along the duct, and apocrine glands, lining the inner wall of the sac itself. The contents of these structures result from the combination of these glandular secretions, to which desquamated keratinocytes and fluid are added. This mixture presents notable individual characteristics in terms of color, consistency, and odor, which explains the macroscopic variability observed from one animal to another during clinical examination.
The physiological emptying of this content occurs through a mechanical mechanism: the pressure exerted by the fecal bolus during transit, combined with the contraction of the perineal musculature at the time of defecation. This physiological mechanism is therefore closely dependent on fecal consistency, perineal muscle tone, and the absence of ductal obstruction. When these mechanisms are insufficient — due to abnormal fecal consistency, muscular tonal deficiency, or local inflammation — the content accumulates and can progress toward increasingly severe pathological states, from impaction to abscess, passing through sacculitis. Understanding this physiology is essential for identifying predisposing factors and preventing recurrences.
The Perineal and Perigenital Regions
The perineal region anatomically corresponds to the diamond-shaped area bounded cranially by the base of the tail and caudally by the base of the external genitalia. Its permanent exposure to urine, fecal matter, and exogenous irritants makes it a particularly vulnerable zone for dermatoses, especially in breeds with reduced hair density in this area. This limited hair protection leaves the epidermis directly exposed to chemical and mechanical agents from the immediate environment, thereby facilitating the development of irritant dermatitis, bacterial or fungal superinfections, and chronic maceration lesions. The perigenital region encompasses the external genitalia of both sexes, with specific clinical entities depending on the sex — vulvar intertrigo in the female, scrotal dermatitis in the male — which will be addressed in dedicated sections. Perigenital tumors, though less common than those of the perianal region, also constitute a differential diagnosis to consider when facing any progressively growing mass in this location.
Semiology and Initial Diagnostic Approach
Interpreting Behaviors: Scooting and Licking
A clinician faced with a dog presented for licking or rubbing the perianal region on the ground must first place these behaviors within their physiological context before concluding that pathology is present. Published data indicate that 24% of clinically healthy dogs spontaneously engage in this “scooting” behavior, while 36% lick the perianal region with a mean intensity rated at 3.5 out of 10. These figures demonstrate normal behavioral variability and call for caution in interpretation. Licking and rubbing of the perianal region can indeed be part of entirely physiological grooming behaviors in the canine species.
It is the increase in frequency and intensity of these behaviors, relative to the animal’s baseline behavior, that should alert the clinician and point toward subclinical or clinical pathology. Excessive licking, increased irritability when the region is handled, or self-mutilation should be considered warning signs warranting thorough examination. The history should systematically clarify the duration of signs, their permanent or intermittent nature, the occurrence of recent digestive episodes — particularly diarrheal episodes — and the known allergic or dermatological history of the animal. This information allows early orientation of the diagnostic approach toward one of the major etiological categories: anal sac conditions, allergic dermatoses, immune-mediated processes, or neoplasias.
Differentiating Anal Sac Disease from Strictly Cutaneous Dermatosis
The first diagnostic step, when faced with these warning signs, is to establish whether the origin is glandular — that is, related to the anal sacs — or purely cutaneous. This distinction determines the entire subsequent approach. Digital rectal examination by internal palpation remains the reference method: it allows assessment of sac symmetry, degree of filling, presence of elicited pain, and content consistency. A superficial examination limited to external observation without internal palpation is insufficient and exposes the clinician to diagnostic errors through inadequate lesion characterization. The pain that is sometimes present should nonetheless be taken into account before proceeding.
One point deserves emphasis, as it is a frequent source of diagnostic errors: macroscopic and microscopic evaluation of anal sac contents is not predictive of disease. Both intracellular and extracellular bacteria, neutrophils, and Malassezia yeast are regularly found in samples from the anal sacs of healthy dogs. Systematic bacteriological culture of the contents, performed without the clinical context of a confirmed abscess, therefore provides no isolated diagnostic value. A clinician who bases their therapeutic decision solely on these cytological or microbiological results would be exposed to prescription errors, notably unjustified antibiotic therapy. Diagnosis fundamentally relies on the demonstration of clinically detectable tissue inflammation and elicited pain on palpation.
Anal Sac Conditions: From Functional to Pathological
Non-neoplastic anal sac conditions represent a frequent cause of consultation in general veterinary medicine, with an annual prevalence estimated at 4.40% in British first-opinion practices. Impaction constitutes the most common form, representing nearly 79% of cases, followed by sacculitis (12%) and abscess (9%). Marked breed predispositions have been documented: brachycephalic types present 2.6 times greater risk of developing anal sac disease than dolichocephalic types. The Cavalier King Charles Spaniel, the King Charles Spaniel, Cockapoos, and Bichon Frisés are among the most at-risk breeds. Conversely, the Labrador Retriever, German Shepherd, Border Collie, and Boxer present reduced risk compared to mixed-breed dogs. These epidemiological data have direct practical implications: breed must be integrated into risk stratification during history-taking, and the systematic identification of underlying allergic disease — notably atopic dermatitis, the most common comorbidity associated with recurrent sacculitis — determines the effectiveness of long-term management.
An Evolutionary Continuum: Impaction, Sacculitis, and Abscess
These three clinical entities do not constitute independent conditions but rather successive stages of the same evolving pathological process. Understanding this continuum is essential for adapting management to each stage and for anticipating potential complications when initial management is insufficient or delayed. Each stage has its own clinical characteristics, specific treatment, and therapeutic limits to be respected.
Anal Sac Impaction
Impaction is defined by the excessive accumulation of thickened or dried content within one or both anal sacs. Clinical signs remain, at this stage, relatively discrete: predominantly perianal pruritus, erythema, and desquamation secondary to pruritus in advanced forms. The sacs are neither inflamed nor painful and express without notable resistance upon palpation. This relative clinical simplicity sometimes contrasts with the functional discomfort experienced by the animal, which may manifest as markedly increased licking or scooting.
Management relies on manual expression of the sacs, a simple procedure whose benefit remains limited if not accompanied by identification and treatment of the underlying cause. Etiological factors to be systematically sought include dietary abnormalities — particularly low-residue feeding that produces small, insufficiently firm stools unable to provide effective mechanical pressure on the sacs — chronic allergic conditions that may alter the quality and quantity of secretions, and repeated episodes of diarrhea that disrupt the physiological emptying mechanism. Antibiotic therapy and analgesia are not indicated at this stage; their prescription constitutes a therapeutic error by excess. Regular scheduled expressions, combined with dietary correction, are generally sufficient to control recurrent impaction when the predisposing cause is correctly identified.
Sacculitis
Sacculitis corresponds to inflammation of the anal sac, most often presenting unilaterally. The clinical evolution follows a characteristic pattern: initially pruritic, the presentation progressively shifts to become painful as inflammation intensifies. Examination reveals perianal edema, visible swelling, marked erythema, and sacs with purulent or hemorrhagic content. Palpation of the sacs is painful, which clearly distinguishes sacculitis from simple impaction and makes manipulation of the region difficult in a non-sedated, awake animal.
Treatment of sacculitis relies on expression of the sacs under sedation — since pain makes manipulation impossible without prior analgesia — followed by catheterization and thorough flushing of the duct with saline solution or an antiseptic solution. A protocol of intrasaccular flushing followed by infusion of a commercial preparation combining a corticosteroid, antibiotic, and antifungal agent, repeated on average two to three times per animal, achieves clinical resolution in the majority of cases without recourse to systemic antibiotic therapy. Topical application of antiseptics and antimicrobials is recommended, justified by the frequent concurrent presence of superficial pyoderma. Systemic or topical analgesia should be prescribed to ensure the animal’s comfort during the inflammatory resolution phase. This protocol should be repeated at biweekly intervals until complete clinical remission is achieved, defined by the disappearance of tissue inflammation, elicited pain, and abnormal content. Since atopic dermatitis is the most frequently associated comorbidity with sacculitis, its systematic investigation must be integrated into the workup for any recurrent condition.
Anal Sac Abscess
The abscess represents the most advanced stage of the inflammatory process. It is characterized by cutaneous fistulation, sometimes accompanied by systemic hypothermia in severe forms. A counter-intuitive clinical finding worth knowing: rupture of the abscess is often accompanied by a decrease in pain perceived by the animal, due to the tissue decompression it produces. This apparent relief should not lead to minimizing the seriousness of the situation or delaying management, as fistulation marks a breach of the cutaneous barrier with risk of bacterial dissemination and contamination of deep tissues.
Management of the abscess involves incision at the point of maximal fluctuation, followed by swabbing for bacteriological culture and sensitivity testing — an indispensable step for guiding any targeted antibiotic therapy. Flushing, topical application of antiseptics and antimicrobials, and the use of anti-inflammatory agents constitute the foundation of treatment. Systemic antibiotic therapy is initiated only upon receipt of sensitivity test results; clinical data nevertheless show that lesion resolution through topical treatments alone frequently renders its use unnecessary. This approach, based on sensitivity testing rather than systematic empirical antibiotic therapy, is consistent with current guidelines for the appropriate use of antibiotics in veterinary medicine.
Anal Sac Neoplasias: A Diagnosis Not to Be Missed
Apocrine Gland Adenocarcinoma of the Anal Sac
Among malignant neoplasias of the perianal region, anal sac adenocarcinoma occupies a predominant position, representing 17% of malignant tumors in this region. This tumor originates in the apocrine glands of the anal sac wall. It preferentially affects older animals, most often beyond 9 years of age. Contrary to what was long asserted in older literature, large-cohort studies have not demonstrated a significant sex predisposition for this tumor: neither sex presents a clearly higher risk than the other, and diagnostic vigilance must be exercised equally in males and females. However, clear breed predispositions are documented; the English Cocker Spaniel is significantly over-represented with a mean relative risk estimated at 7.3 compared to mixed-breed dogs. Its particularly aggressive biological behavior, combined with the frequency of its incidental discovery, makes it one of the veterinary neoplasias that most imperiously justifies systematic rectal examination at every consultation.
The clinical presentation is dominated by signs that are often unilateral: perianal swelling detectable on palpation, tenesmus, dyschezia, bleeding, and increased licking or scooting behaviors. Bilateral involvement remains possible. Notably, a substantial proportion of these tumors is discovered incidentally during a clinical examination performed for another reason, with the mass remaining asymptomatic at the time of diagnosis. This observation illustrates the necessity of a systematic examination of the perianal region at every consultation, regardless of the initial indication. An animal presented for routine vaccination, dental check, or any other reason should benefit from rectal palpation if their age and clinical profile justify it.
The Paraneoplastic Syndrome of Hypercalcemia
Apocrine gland adenocarcinoma of the anal sac has the capacity to secrete a parathyroid hormone-related protein (PTHrP). This ectopic secretion induces a paraneoplastic syndrome of humoral hypercalcemia, reported in 25 to 90% of cases depending on the published cohort, and may manifest as polyuria-polydipsia, muscle weakness, constipation, or cardiac arrhythmias. This syndrome resolves after complete tumor excision, making it both a diagnostic marker and a post-surgical control criterion. In some cases, the systemic signs related to hypercalcemia may precede or overshadow the local perianal signs, which may lead to an initial differential diagnosis oriented toward primary hyperparathyroidism or another cause of hypercalcemia. The presence of hypercalcemia also constitutes an independent unfavorable prognostic factor, in addition to exposing the patient to increased cardiovascular and renal anesthetic and surgical risks. Measurement of serum PTHrP, combined with PTH assay and total and ionized calcemia, allows clarification of the biological picture.
Calcium measurement is therefore obligatorily integrated into the preoperative workup, alongside complete biochemistry, urinalysis, and medical imaging aimed at evaluating local and distant disease extension.
Metastatic Potential and Prognostic Factors
The metastatic rate of apocrine gland anal sac adenocarcinoma is high, even for tumors of modest size, making it one of the most treacherous veterinary neoplasias. Dissemination follows a relatively predictable pattern: initial involvement of the regional iliac or sacral lymph nodes, followed by secondary spread to the lungs, liver, and spleen first, as well as to bones and other abdominal organs in advanced stages. Metastases to the vertebral canal have also been documented. The rate of lymph node metastases at presentation varies across cohorts from 26% to more than 90%, depending on imaging methods and inclusion criteria. This variability underscores that even small tumors are not exempt from risk: approximately 20% of animals with a primary tumor smaller than 2 cm already have lymph node metastases at the time of diagnosis. This finding mandates a complete staging workup regardless of tumor diameter at the time of discovery. Abdominal ultrasound and thoracic radiography constitute first-line imaging examinations, while computed tomography offers superior sensitivity for detection of small iliosacrolumbar lymph node metastases, particularly for intrapelvic nodes inaccessible to ultrasonography.
Cytologically, fine-needle aspiration reveals a characteristic neuroendocrine appearance, with bare nuclei and polymorphic atypia. This cytological diagnosis must be confirmed by histopathology. Certain histological parameters have independent prognostic value: a solid growth pattern, marked peripheral infiltration, the presence of necrosis, and lymphovascular invasion are associated with an unfavorable prognosis. Median survival time varies considerably depending on treatment and disease stage. In the absence of any treatment, median survival is approximately three months. Surgery alone achieves a median survival of approximately one year, while combining surgery with adjuvant chemotherapy or radiation therapy can significantly extend this survival, with some multimodal strategies associated with durations exceeding two years. Prognosis is significantly improved when the mass measures less than 2.5 cm at the time of diagnosis, in the absence of detectable metastases, and when resection of the regional lymph nodes is performed concurrently with tumor excision. These prognostic elements underscore the importance of early detection and complete, well-planned surgery. Stereotactic body radiation therapy (SBRT) represents a validated therapeutic option for the treatment of metastatic iliosacrolumbar lymph nodes when surgical resection is declined or not feasible, with high local control rates and median survival comparable to surgical approaches in recent series.
Other Neoplasias of the Anal Sac Region
Squamous cell carcinoma of the anal sac glands, although rare, constitutes an additional serious condition. Its aggressive local behavior and metastatic potential make it an entity not to be overlooked when facing any atypical perianal lesion. Among other less frequent neoplasias affecting the perianal region in the broader sense, one can list apocrine adenomas, various mesenchymal tumors, and mast cell tumors. The latter, in particular, can present in a clinically benign manner in this location, which justifies the systematic use of fine-needle aspiration for any perianal nodular lesion. Each of these entities requires a cytological or histological diagnostic approach for proper characterization and management.
Perianal Conditions Independent of the Anal Sacs
Perianal Erythema and Pruritus: Think Allergy
Pruritic erythema localized to the perianal zone or extending to the entire perineum represents one of the most frequent clinical manifestations in veterinary dermatology. In this context, allergic diseases play a leading role: flea allergy dermatitis (FAD), food allergy, and atopic dermatitis share causality. This clinical sign can, in some cases, constitute the only visible manifestation of allergic disease, without any associated generalized cutaneous involvement. A dog presenting with isolated perianal erythema, without any other identifiable cutaneous lesion, should therefore benefit from a complete allergological investigation rather than simple local symptomatic treatment.
The differential diagnosis includes parasitic infestations — fleas, intestinal parasites such as Dipylidium or hookworms — and Malassezia superinfections, yeast whose proliferation is often secondary to an allergic background. The diagnostic approach progresses by methodical exclusion: infectious and parasitic causes are ruled out first, before undertaking allergological investigation, including a strict elimination diet when food allergy is suspected. The frequent diagnostic wandering observed in these cases often results from incomplete investigation or a poorly conducted elimination diet, underscoring the importance of a structured and methodical approach.
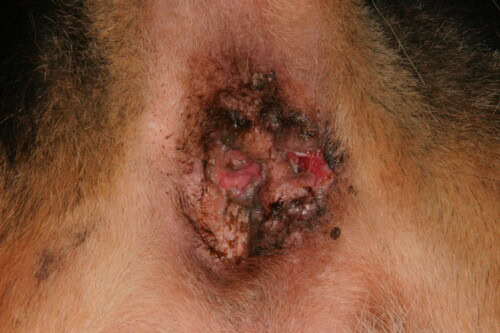
Canine Perianal Fistula: A Chronic Immune-Mediated Disease
Canine perianal fistula, also called anal furunculosis, represents one of the most severe and challenging perianal conditions to manage. It falls within the framework of chronic inflammatory diseases of immune origin, driven by a T-lymphocyte-mediated inflammatory mechanism associated with a tissue healing defect. This healing defect constitutes a major aggravating factor, as it maintains the patency of the fistulous tracts and compromises spontaneous lesion resolution, even in the absence of active bacterial superinfection. The German Shepherd represents more than 80% of reported cases in the literature, reflecting a strong genetic component in susceptibility to the disease. Other breeds may be affected, notably Retrievers and certain breeds with a wide, low-carried tail that promotes a humid, poorly ventilated microenvironment around the anus.
The immunopathological bases of the disease are better characterized today. Allelic variations have been identified in affected German Shepherds. Histopathological analysis of lesions reveals a mononuclear cellular infiltrate dominated by CD3+ T lymphocytes with a cytokine profile compatible with a Th1-type response, notably marked by increased expression of IL-2 and IFN-γ mRNAs in lesional tissues. Overexpression of matrix metalloproteinases MMP-9 and MMP-13 in lesional skin explains the tissue healing defect that maintains the fistulous tracts. A dysfunction of the NOD2 receptor, involved in the recognition of bacterial pathogen-associated molecular patterns, has also been proposed as a contributing mechanism. Furthermore, significant dysbiosis of the cutaneous and rectal microbiota has been documented in affected German Shepherds, with significant compositional differences compared to healthy dogs. This dysbiosis changes during lesion resolution under immunomodulatory treatment, opening perspectives on the potential role of the microbiome in the pathogenesis of the disease. Taken together, these data position canine perianal fistula as a spontaneous animal model of fistulizing Crohn’s disease in humans, with which it shares genetic and immunopathological bases.
Moderate perianal fistulas
Clinical Presentation
The disease is morphologically characterized by multiple fistulous tracts and ulcers of highly variable size, ranging from discrete millimeter-sized lesions to massive, destructive ulcerations. The typically circumferential distribution of these lesions around the anus constitutes a discriminating semiological element, allowing it to be distinguished from focal involvement of the anal sacs. This circumferential distribution reflects diffuse involvement of the perianal cutaneous zone and not a focal lesion related to obstruction of a sac. Functional clinical signs include compulsive licking and scooting, painful dyschezia that may lead to behavioral anorexia — the animal refusing to eat in anticipation of the pain associated with defecation — as well as the presence of blood and exudates around the anal region.
Concurrent involvement of the anal sacs or rectum remains possible and must be actively sought when therapeutic response proves insufficient. Endoscopic exploration may then be necessary to evaluate the extent of rectal mucosal involvement and guide the therapeutic decision. Concurrent colitis has been reported in some cases, suggesting a continuum between canine perianal fistula and chronic inflammatory bowel disease, similar to what is observed in Crohn’s disease in humans. Furthermore, an association between food reactivity and perianal fistula has been documented in the German Shepherd, justifying the addition of an elimination diet to immunomodulatory treatment in cases of frequent relapses despite well-conducted immunosuppression.
Therapeutic Management
One fundamental point must be firmly established in clinical practice: antibiotics are ineffective on the progression of the underlying disease. Their isolated or prolonged use does not modify the underlying immune-mediated process — T-lymphocyte inflammation, overexpression of MMP-9 and MMP-13, healing defect — and exposes the patient to the risk of bacterial resistance. This point is all the more important because canine perianal fistula is a disease with frequent relapses, and repeated empirical antibiotic therapy at each recurrence constitutes a harmful long-term therapeutic drift. The reference treatment relies on immunomodulators. Oral cyclosporine constitutes the first-line molecule, with a high level of evidence: administered until clinical remission is achieved, the dose is then progressively reduced to the minimum effective threshold. The combination of cyclosporine and ketoconazole represents a validated pharmacological strategy that allows, through hepatic enzymatic inhibition, an increase in cyclosporine bioavailability and a reduction of more than 80 to 90% in the dose needed to achieve therapeutic blood concentrations. This combination significantly reduces the cost of treatment in countries where ketoconazole is inexpensive — which is not the case in France — without compromising efficacy, and should be considered as first-line when the owner’s budget is a constraint. In case of failure of cyclosporine alone, the second-line strategy recommended by the most recent literature review is the combination of systemic prednisolone and topical tacrolimus, not tacrolimus alone. Topical tacrolimus represents an effective local alternative, particularly suited to moderate-sized lesions when used in this combined framework. It should be noted that the use of topical tacrolimus is prohibited in some countries, such as France. French veterinarians can therefore neither legally prescribe nor obtain this medication.
Other therapeutic options have been reported in the literature with varying levels of evidence: corticosteroids, azathioprine, oclacitinib, mycophenolate mofetil, fluorescent light therapy, and dietary modifications. The latter deserve particular attention, as a predisposing dietary component is suspected in some cases, leading some clinicians to combine an elimination diet with immunomodulation. Oclacitinib, an inhibitor of the JAK-STAT pathway involved in the signaling of pro-inflammatory interleukins, represents a promising therapeutic avenue whose clinical evaluation has yielded favorable results in cases resistant to cyclosporine. Fluorescent light therapy (photobiomodulation), through its anti-inflammatory and biostimulating effects on tissue healing, also constitutes an interesting adjunct tool, notably for promoting the closure of resistant fistulous tracts. Cell therapies based on mesenchymal stem cells represent an experimental avenue currently under exploration.
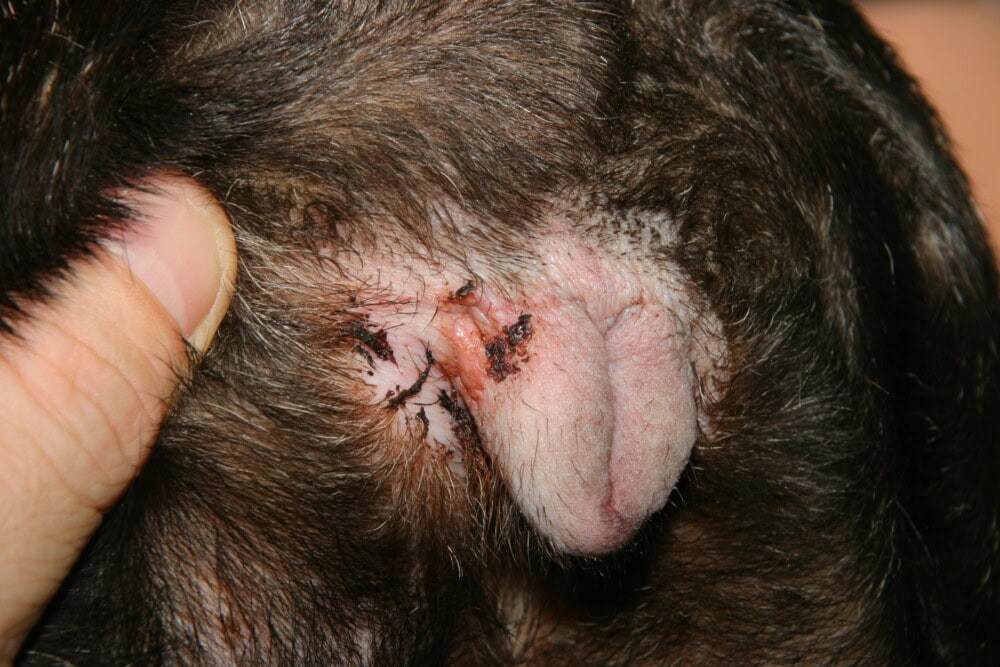
Hepatoid Gland Tumors: A Distinct Entity
Hepatoid glands — named for the histological resemblance of their cells to hepatocytes — are modified sebaceous glands confined to the perianal cutaneous zone. The tumors arising from them collectively represent 25% of all canine cutaneous tumors, making them a quantitatively major neoplastic group and one of the most frequently encountered in veterinary dermatological oncology.
Hepatoid Adenoma
Hepatoid adenoma is the most common form, appearing as solitary or multiple masses, with a marked predilection for intact middle-aged to older males. This sex predisposition reflects a hormone-dependent pathogenesis, with androgens playing a trophic role on these glands. Nodular perianal hyperplasia of hepatoid glands, observed in intact middle-aged males, may precede the development of a true adenoma; this benign nodular hyperplasia also constitutes an indication for castration. Diagnosis is established on the basis of clinical examination — sessile or pedunculated, fleshy mass, sometimes with an ulcerated surface — and confirmed by cytology or histology. Presurgical cytology provides useful information for guiding the operative decision: several cytological criteria allow differentiation of benign from malignant lesions with satisfactory diagnostic accuracy, without however replacing histopathological examination.
Curative treatment systematically combines complete surgical excision of the mass and castration — surgical or chemical — the latter being indispensable to prevent tumor recurrences related to residual androgenic stimulation. Castration alone can induce partial regression of some hepatoid adenomas, but surgical excision remains the reference treatment to eliminate the in-place lesion and obtain a definitive histological diagnosis. The prognosis is excellent, with metastases remaining exceptionally rare for adenoma. This favorable prognosis contrasts with that of hepatoid adenocarcinoma and underscores the importance of precise histological characterization for correctly orienting management.
Hepatoid Adenocarcinoma
Hepatoid adenocarcinoma is considerably rarer. Its more aggressive biological behavior translates into a more marked local invasion potential and a higher metastatic risk than that of adenoma. This behavioral difference mandates a more rigorous diagnostic and surgical approach, with an appropriate staging workup — including abdominal and thoracic imaging — before any operative decision. The histological distinction between hepatoid adenoma and adenocarcinoma can prove challenging in some cases and requires the expertise of an experienced veterinary pathologist. Histological grading systems have been proposed to refine prognostic stratification beyond the simple benign/malignant dichotomy, integrating architectural and cytological criteria capable of predicting clinical outcome.
Dermatoses of the Perineal Region
The Diagnostic Framework: History and Chronology
The perineal region, due to its anatomical location between the anus and the genitalia, undergoes chronic exposure to fecal matter, urine, and various irritants. Breeds with low perineal hair density are particularly predisposed to dermatoses in this area. When facing a perineal lesion, the diagnostic approach relies on a precise history to determine the chronology of lesion appearance and progression. Three clinical frameworks are clearly distinguished: acute presentation, chronic presentation, and nodular presentation, each pointing toward a distinct etiological spectrum and requiring an adapted diagnostic strategy.
Acute Presentation: Irritation as a Triggering Factor
An acute presentation typically occurs in the context of a recent episode of diarrhea or fecal or urinary incontinence. Clinical examination reveals erythematous, moist skin with variable exudation. The primary differential diagnosis points toward irritant contact dermatitis — with the digestive enzymes contained in diarrheal stools constituting the main injurious agent, as proteases and bile salts exert a directly corrosive action on the perineal epidermis — and toward pressure sores in cases of prolonged immobility. Secondary bacterial superinfection must always be investigated by cytology, as exudative erythemas constitute a favorable environment for the proliferation of cocci and bacilli.
Chronic Presentation: The Dominant Allergic Background
In its chronic form, perineal dermatosis manifests as persistent pruritus and licking. Advanced lesions present a characteristic cutaneous picture: erythema, friction-induced alopecia, hyperpigmentation, lichenification, and marked seborrhea. These changes reflect chronic cutaneous remodeling related to self-perpetuating pruritus, in which repeated scratching and licking impair the cutaneous barrier and promote the penetration of allergens and infectious agents. This pruritus-lesion-pruritus vicious cycle is central to the pathophysiology of chronic allergic dermatitis.
The differential diagnosis primarily points toward an underlying allergic dermatitis — atopic, food-related, or flea bite-related — frequently complicated by bacterial pyoderma or Malassezia dermatitis. These secondary infections contribute to maintaining the pruritus-scratching-lesion cycle and must be treated concurrently with the primary cause. A diagnostic approach that treats only secondary infections without identifying and controlling the underlying allergic disease is doomed to fail and leads to frequent relapses.
Nodular Presentation: Neoplasias and Granulomas as Priorities
Progressively growing perineal nodular lesions that are initially non-pruritic constitute a clinical presentation requiring rapid cytological or histological investigation. The differential diagnosis must include various cutaneous neoplasias, infectious nodules — deep bacterial fistulas or mycotic infections — and parasitic granulomas. Fine-needle aspiration represents the first recommended diagnostic procedure for any perianal or perineal nodular lesion. This simple procedure, performable during consultation without sedation in most cases, allows rapid orientation toward an inflammatory, infectious, or neoplastic nature and enables planning of subsequent management accordingly. Biopsy with histopathological examination remains indispensable when cytological results are inconclusive or when precise tissue characterization is necessary for treatment decision-making.
Perigenital Conditions: Sex-Specific Features
In the Female: Vulvar Intertrigo
Vulvar intertrigo is a common dermatological condition in female dogs, particularly observed in the presence of two major predisposing factors: obesity and hypoplastic vulva. In these anatomical configurations, the perivulvar skin folds create a warm, humid, macerated microenvironment conducive to the accumulation of secretions — vaginal secretions, residual urine, cellular debris. This accumulation promotes the development of local inflammatory reactions, erosions, and secondary bacterial or fungal superinfections. The pathogens most frequently implicated in vulvar intertrigo superinfections include gram-positive bacteria such as staphylococci and Malassezia yeast. Associated clinical signs include perivulvar erythema, exudation, local pruritus, and sometimes a malodorous smell. Management ideally combines local treatment of the infection and correction — when possible — of the anatomical or weight-related predisposing factor. Perivulvar surgery — episioplasty or vulvoplasty — may be considered to anatomically correct excessive folds in female dogs with severe hypoplastic vulva and frequent recurrences despite well-conducted medical management.
Vulvar intertrigo
In the Male: Scrotal Dermatitis
The scrotum, due to the thinness and sensitivity of its epidermis, represents a predilection zone for inflammatory dermatological reactions in male dogs. The particular thinness of the scrotal epidermis, combined with its direct exposure to the environment, makes it highly reactive to allergic, irritant, and infectious stimuli. Scrotal dermatitis is frequently observed, with an etiological spectrum including allergic reactions — atopy, contact allergy — bacterial infections, and Malassezia dermatitis. Contact with irritant substrates, household products, or rough surfaces can also trigger or aggravate this presentation, particularly in animals in prolonged contact with chemically treated surfaces. Cutaneous cytology rapidly points toward the pathogen(s) involved and guides topical treatment. Recurrent scrotal dermatitis should systematically raise suspicion of underlying atopic dermatitis, of which it may constitute one of the predominant clinical manifestations.
Perigenital Tumors
Tumors affecting the perigenital region are less common than those of the perianal region but deserve consideration in the differential diagnosis of any progressively growing perigenital mass. Among the perigenital neoplasias encountered in dogs, one can mention transmissible venereal tumors, squamous cell carcinomas, and various mesenchymal tumors. The transmissible venereal tumor, although still uncommon in Western Europe, should appear in the differential diagnosis of perigenital masses in dogs with a history of travel to enzootic areas. Surgical treatment generally constitutes the first therapeutic intention for benign or localized perigenital tumors, while malignant forms may require a combined approach associating surgery, chemotherapy, or radiation therapy depending on histotype and stage.
Summary
The management of perianal, perineal, and perigenital dermatological conditions in dogs relies on a rigorous clinical approach, articulated around three complementary axes. First, precise knowledge of regional anatomy, which determines the recognition of structures involved and the interpretation of observed lesions. Second, a refined semiology distinguishing acute from chronic conditions, pruritic from painful presentations, and anal sac conditions from independent cutaneous dermatoses. Third, judicious use of complementary examinations — cytology, targeted bacteriological culture, imaging, biochemical workup — whose diagnostic value must be interpreted in clinical context and not in isolation.
The algorithmic approach based on lesion chronology — acute or chronic — constitutes a particularly valuable structuring tool for the clinician faced with the diversity of perineal clinical presentations. This approach allows hierarchization of the differential diagnosis, selection of the most relevant complementary examinations, and avoidance of unnecessary investigations or non-targeted empirical treatments.
Two cross-cutting lessons deserve particular attention. On one hand, systematic bacteriological culture of anal sac contents does not constitute a reliable diagnostic tool in the absence of the clinical context of a confirmed abscess, due to the habitual presence of microorganisms in this compartment in healthy animals. On the other hand, systemic antibiotic therapy must be reserved for situations where it is genuinely justified — abscess with sensitivity testing, documented superinfection — and must under no circumstances constitute an empirical first-line treatment for uncomplicated inflammatory presentations.
Research perspectives in this field notably concern understanding the precise immunopathological mechanisms of canine perianal fistula, whose complexity remains partially elucidated — particularly the role of the perianal microbiota as a factor potentially modifiable by treatment — as well as the identification of predictive biomarkers of recurrence for hepatoid tumors. The prospective evaluation of new immunomodulatory molecules, such as JAK inhibitors applied to perianal fistula, opens promising therapeutic avenues that will deserve documentation through controlled clinical trials. Furthermore, improvement of preoperative staging techniques for apocrine gland anal sac adenocarcinoma — notably through systematic recourse to computed tomography and development of molecular markers predictive of metastatic potential — as well as evaluation of stereotactic body radiation therapy in multimodal treatment, constitute active research axes likely to significantly improve the prognosis of this tumor.
Conclusion
The perianal, perineal, and perigenital regions concentrate, within a restricted anatomical territory, a remarkable diversity of dermatological conditions whose etiologies, mechanisms, and treatments differ fundamentally. These regions play an important role in canine social communication, which fully justifies the clinical attention devoted to them beyond their purely medical importance. Systematic rectal examination, rigorous analysis of lesion chronology, and judiciously performed cytology allow precise diagnostic orientation. The distinction between functional anal sac disease, immune-mediated inflammatory process, allergic dermatosis, and neoplasia directly determines the effectiveness of therapeutic management. Neglecting this region or reducing it to a superficial examination exposes the clinician to diagnostic delays with sometimes serious clinical consequences — notably in the case of apocrine gland anal sac adenocarcinoma, a tumor with no established sex predisposition, affecting all older animals regardless of their sex, and whose high metastatic potential renders every week of diagnostic delay potentially detrimental to the prognosis.
Maina E. From perianal to perigenital conditions in dogs. Practical program of the 35th European Veterinary Dermatology Congress. Bilbao, Spain; September 11–13, 2025.