Canine atopic dermatitis affects 10 to 15% of the global canine population and is one of the leading reasons for veterinary dermatology consultations. Faced with this widespread condition, new treatments are emerging with an excellent benefit/risk ratio. Here is a comprehensive overview of Zenrelia (ilunocitinib), the first of the new Janus Kinase inhibitors to have been marketed after oclacitinib (Apoquel).
PART I — INTRODUCTION AND CONTEXT
1.1 Canine atopic dermatitis: nosological framework
1.1.1 Definition and pathophysiology
Canine atopic dermatitis (CAD) is a chronic inflammatory skin disease with a genetic predisposition, characterized by recurrent pruritus and erythematous skin lesions associated with sensitization to environmental allergens (Hensel 2015). The pathophysiology of CAD rests on three intertwined pillars: epidermal barrier dysfunction, immune dysregulation, and progressive allergen sensitization. The epidermal barrier is altered by structural abnormalities of the stratum corneum, comparable to those described in human atopic dermatitis where filaggrin gene mutation is documented. In dogs, studies have highlighted abnormal expression of intercellular junction proteins (claudins, occludin) in lesional skin biopsies from atopic dogs (Marsella 2021). This barrier disruption facilitates transcutaneous penetration of allergens and microorganisms, sustaining chronic antigenic stimulation.
From an immunological standpoint, CAD is characterized by an initial Th2-type polarization with overexpression of interleukins IL-4, IL-5, IL-13, and IL-31, followed by a chronic phase in which Th1 responses (interferon-gamma, IFN-γ) and Th17 responses coexist (Pucheu-Haston 2015). IL-31, a major pruritogenic cytokine, binds to its heterodimeric receptor (IL-31RA/OSMR) expressed on cutaneous sensory neurons and activates the JAK1/JAK2-STAT3 signaling pathway, generating the itch signal transmitted to higher nerve centers (Gonzales 2013). Significant elevations in circulating concentrations of IL-31, IL-17, and total IgE have been documented in atopic dogs compared to healthy controls (Chaudhary 2019). Thymic stromal lymphopoietin (TSLP) and substance P contribute to the amplification of this pruritogenic cascade. The role of the skin microbiome, with proliferation of Staphylococcus pseudintermedius at lesional sites, is a documented aggravating factor that sustains the inflammatory loop through activation of keratinocyte Toll-like receptors (TLR-2) (Santoro 2015).
1.1.2 Epidemiology and breed predispositions
The prevalence of CAD is estimated between 10 and 15% of the global canine population, with regional variations related to environmental conditions and breeding practices (Hillier 2001). Approximately 17 million dogs suffer from allergic skin diseases in the United States, including CAD, food allergies, and flea allergy dermatitis (Drechsler 2024). CAD is one of the leading reasons for veterinary dermatology consultations and represents a considerable economic burden for pet owners, with annual treatment costs estimated between $500 and $2,000 per animal, depending on the country, therapeutic protocol, and severity.
Several breeds present a well-documented genetic predisposition. The Labrador Retriever manifests a clinical phenotype dominated by bilateral and symmetrical erythematous pruritus of the palmar and plantar interdigital spaces, ear pinnae, and axillary folds, with recurrent ceruminous external otitis and chronic pododermatitis. The West Highland White Terrier develops an early and severe form of CAD, with high prevalence in certain lines, characterized by intense facial, periocular, and perilabial erythema, associated with rapid lichenification of flexion areas (Favrot 2020). The French Bulldog presents clinical manifestations with inguinal, perineal, and interdigital tropism, with an increased frequency of bacterial and fungal (Malassezia pachydermatis) superinfections requiring concurrent anti-infective treatments. The Golden Retriever, Boxer, Shar Pei, and German Shepherd are among the other high-risk breeds (Hensel 2015). The Shar Pei deserves special mention: the severity of CAD in this breed correlates with excessive dermal mucin, which impairs cutaneous thermoregulation and promotes maceration in skin folds, creating an environment conducive to superinfections. The Boxer presents a particular clinical profile with predominant ventral abdominal and inguinal involvement, early lichenification, and a propensity for proliferative otitis. No specific susceptibility gene for CAD has been formally identified in dogs, unlike ichthyosis in the Golden Retriever (PNPLA1 mutation).
The Jack Russell Terrier is one of the many breeds predisposed to canine atopic dermatitis
1.2 Therapeutic options available before ilunocitinib
1.2.1 Glucocorticoids
Systemic glucocorticoids (prednisolone, methylprednisolone) have long been the first-line treatment for CAD due to their rapid efficacy on pruritus and inflammation. Prednisolone provides a reduction in pruritus of more than 50% in approximately 70% of dogs within the first 48 hours (Olivry 2015). However, prolonged use exposes patients to well-characterized systemic adverse effects: polyuria-polydipsia, polyphagia, weight gain, cutaneous atrophy, calcinosis, iatrogenic diabetes mellitus, increased susceptibility to urinary and cutaneous infections, and suppression of the hypothalamic-pituitary-adrenal axis.
1.2.2 Cyclosporine
Cyclosporine, a calcineurin inhibitor, was the first immunomodulatory molecule to obtain a marketing authorization specifically for CAD. At a dose of 5 mg/kg/day orally, it reduces pruritus by 50% in approximately 65% of dogs after 4 to 6 weeks of treatment (Steffan 2006). Its prolonged onset of action (4 to 6 weeks to reach maximum efficacy) and the frequency of digestive side effects (vomiting in 25 to 30% of animals at the start of treatment) limit therapeutic compliance.
1.2.3 Oclacitinib
Oclacitinib (APOQUEL, Zoetis), the first JAK inhibitor marketed in veterinary medicine in 2013, represented a major therapeutic advance. This preferential inhibitor of JAK1 and JAK3 inhibits IL-31, IL-4, and IL-13 signaling, providing rapid reduction in pruritus as early as 4 hours after administration (Gonzales 2014). The recommended dosage is 0.4–0.6 mg/kg twice daily for 14 days (induction phase), then once daily as maintenance therapy. The study by Cosgrove et al. (Cosgrove 2015) documented satisfactory efficacy and tolerability over 630 days of continuous use. However, the transition from twice-daily to once-daily administration during the maintenance phase exposes patients to a pruritus rebound phenomenon between day 14 and day 28, documented by Fukuyama et al. (2017) and Olivry et al. (2023).
1.2.4 Lokivetmab
Lokivetmab (CYTOPOINT, Zoetis), a caninized anti-IL-31 monoclonal antibody, represents the first targeted biotherapy in veterinary dermatology. It is administered by subcutaneous injection at a minimum dose of 1 mg/kg to 2 mg/kg every 4 to 8 weeks. The initial dose-response trial (Michels 2016) evaluated doses of 0.125 to 2 mg/kg. It specifically neutralizes circulating IL-31 and provides pruritus relief in approximately 70% of treated dogs (Michels 2016). Its purely antipruritic mode of action, without direct anti-inflammatory activity on skin lesions, and the need for monthly in-clinic injections are its limitations. The lokivetmab–oclacitinib combination has been described in the literature for refractory cases.
1.3 Development history and regulatory approval
1.3.1 Preclinical and regulatory development
Ilunocitinib was developed by Elanco Animal Health as a new molecular entity in the class of JAK inhibitors, specifically designed for veterinary use in the treatment of pruritus associated with canine allergic dermatitis. The principal patent describes the molecule as compound 27 in a series of azetidine and cyclobutane derivatives active on Janus kinases, with a projected expiration in 2040 (patent US 11548895). The clinical development program included target species safety studies (Kuntz 2025), pharmacokinetic studies (Boerngen 2026), three field clinical trials (Forster 2025a, 2025b, 2025c), and two vaccine response studies (Fent 2025a, Fent 2025b).
The initial marketing authorization was granted by the US Food and Drug Administration (FDA) on September 19, 2024, for the control of pruritus associated with allergic dermatitis and the control of atopic dermatitis in dogs aged at least 12 months. Commercial launch in the United States immediately followed this approval. Subsequently, authorization was extended to Brazil, Canada (December 2024), and Japan during the first half of 2025. In Europe, the Committee for Medicinal Products for Veterinary Use of the European Medicines Agency (EMA) adopted a positive opinion in June 2025, and the European marketing authorization was granted by the European Commission in July 2025, with an immediate commercial launch. In France, the veterinary medicinal product ZENRELIA has been available by veterinary prescription since the third quarter of 2025. In September 2025, the FDA approved an update to the US label, removing the mention of “fatal vaccine-induced disease” from the labeling, based on additional scientific data demonstrating that the cases of adenoviral hepatitis observed in the initial vaccine study resulted from natural infection with CAV-1, and not from vaccine-induced disease (Fent 2025a). More than 800,000 dogs have been treated worldwide since launch, according to data communicated by Elanco at the end of 2025.
PART II — CHEMISTRY AND MOLECULAR STRUCTURE
2.1 Chemical identity and physicochemical properties
2.1.1 Molecular structure
The international nonproprietary name of the molecule is ilunocitinib. The brand name is Zenrelia (Elanco). The molecular formula is C₁₇H₁₇N₇O₂S, corresponding to an exact molar mass of 383.12 Da and a molecular weight of 383.43 g/mol. Ilunocitinib belongs to the pyrrolopyrimidine family with azetidine and sulfonamide functional groups, structurally distinct from oclacitinib, another pyrrolopyrimidine but bearing a trans-substituted cyclohexylamine group and an N-methylated methanesulfonamide. Ilunocitinib shares with baricitinib a common mechanism of action (competitive inhibition of the ATP site of JAK) and a partially overlapping isoform selectivity profile (JAK1/JAK2), but belongs to a structurally distinct chemical family (Clark 2014). The three-dimensional configuration of ilunocitinib confers occupation of the ATP-dependent catalytic site of JAK1, JAK2, and TYK2 kinases, with differential affinity depending on the isoform.
2.1.2 Physicochemical properties
Ilunocitinib exhibits low aqueous solubility, a limiting factor for absorption at high doses, and moderate to high permeability in the Caco-2 cell model, with apparent permeability (Papp) values ranging from 6.74 × 10⁻⁶ to 17.45 × 10⁻⁶ cm/s (Boerngen 2026). This characteristic classifies the molecule in the Biopharmaceutics Classification System (BCS) as class II or IV depending on experimental conditions. Ilunocitinib is a substrate of gastrointestinal efflux transporters, with functional homology to P-glycoprotein (P-gp), a property shared with baricitinib and tofacitinib in human medicine (Spinelli 2021).
2.2 Pharmaceutical formulation
2.2.1 Film-coated tablets and dosages
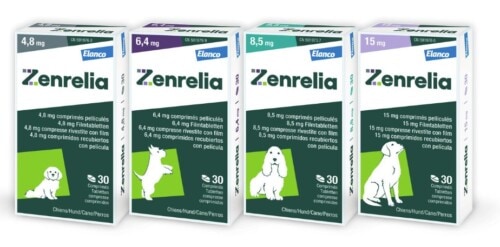
The film-coated tablets of Zenrelia are available in four dosage strengths: 4.8 mg, 6.4 mg, 8.5 mg, and 15 mg of ilunocitinib. Each tablet is scored, allowing a combination of multiple tablets to adjust the dose to the animal’s body weight. Ilunocitinib tablets are packaged in blisters of 10 and 30 units and in bottles of 90 units. The hypoallergenic, non-flavored formulation allows administration with or without food, without a significant effect on efficacy under real clinical conditions (Boerngen 2026).
PART III — MECHANISM OF ACTION AND PHARMACODYNAMICS
3.1 The JAK-STAT pathway: fundamental overview
3.1.1 Architecture of the signaling cascade
The JAK-STAT (Janus Kinase – Signal Transducer and Activator of Transcription) signaling pathway is one of the most conserved cytokine signal transduction mechanisms throughout the evolution of metazoans. This pathway transmits signals from type I and type II membrane cytokine receptors to the cell nucleus, where it regulates transcription of genes involved in immunity, inflammation, hematopoiesis, and cell proliferation (O’Shea 2015). The JAK kinase family comprises four members: JAK1, JAK2, JAK3, and TYK2 (tyrosine kinase 2). Each JAK kinase consists of seven homology domains (JH1 to JH7), with the JH1 domain carrying tyrosine kinase catalytic activity and the JH2 domain exerting a regulatory pseudokinase function. The STAT transcription factor family comprises seven members (STAT1 to STAT4, STAT5a, STAT5b, STAT6).
The transduction mechanism follows an ordered sequence: cytokine binding to its membrane receptor induces receptor dimerization, causing the approximation and trans-phosphorylation of JAK kinases associated with the intracytoplasmic portion of the receptor. Activated JAKs then phosphorylate tyrosine residues on the receptor, creating docking sites for STAT proteins. The recruited STATs are in turn phosphorylated by the JAKs, dimerize, and translocate to the nucleus where they bind to specific response elements on genomic DNA, activating or repressing transcription of target genes (Spinelli 2021). Pharmacological inhibition of this cascade by JAK inhibitors (JAKi) simultaneously blocks the signaling of multiple cytokines sharing the same JAK pairs, which explains both the therapeutic efficacy and the adverse effect profile of this drug class.
3.2 Selectivity of ilunocitinib for JAK subtypes
3.2.1 Isoform selectivity profile
Ilunocitinib is described as a “non-selective” JAK inhibitor, with a selectivity profile characterized by high in vitro inhibitory potency against JAK1, JAK2, and TYK2, and lesser activity against JAK3 (Forster 2025c). This selectivity distinguishes it from oclacitinib, which preferentially acts on JAK1 and JAK3 (Gonzales 2014). Inhibition of JAK1 is the mechanism shared by both molecules, responsible for blocking IL-4 signaling (via the JAK1/JAK3 complex), IL-13 signaling (via JAK1/TYK2 or JAK1/JAK2), and IL-31 signaling (via JAK1/JAK2). Inhibition of JAK2 by ilunocitinib implies a potential impact on the erythropoietin pathway (EPO/JAK2/STAT5), explaining the dose-dependent hematological changes observed in the safety study (Kuntz 2025). Inhibition of TYK2 blocks IL-12 and IL-23 signaling (TYK2/JAK2-STAT4 pathway), involved in Th1/Th17 polarization, adding a complementary anti-inflammatory dimension. The minimal impact on other protein kinases and lipid kinases limits the risk of off-target effects (Choy 2019, Bonelli 2024). However, in vitro enzymatic selectivity data do not directly predict the clinical efficacy and tolerability of atopic dermatitis treated with JAKi, as Virtanen et al. (Virtanen 2023) reminded in their comparative analysis of isoform selectivity profiles of JAKi used in human rheumatology. Head-to-head comparative clinical trials remain the gold standard for comparison between molecules.
3.3 Targeted cytokines
3.3.1 Spectrum of cytokine inhibition
Inhibition of JAK1, JAK2, and TYK2 kinases by ilunocitinib blocks the signaling of a broad spectrum of cytokines involved in the pathogenesis of CAD. Among the directly inhibited pruritogenic cytokines are IL-31 (the major mediator of canine atopic pruritus, acting via the IL-31RA/OSMR receptor complex and the JAK1/JAK2 pair), TSLP (thymic stromal lymphopoietin, an epithelial alarm cytokine activating the JAK1/JAK2-STAT5 pathway), and, indirectly, substance P, whose neuronal expression is modulated by IL-31-dependent signals (Datsi 2021). From a neuroanatomical standpoint, the IL-31RA/OSMR receptor complex expressed on cutaneous sensory neurons transmits the pruritogenic IL-31 signal to the dorsal root ganglia, where relay to the ascending spinothalamic pathways leading to central itch perception occurs (Nemmer 2021). This anatomical precision explains the rapid onset of action of JAKi on pruritus: inhibition of JAK1/JAK2 phosphorylation at the peripheral sensory neuron level interrupts the signaling cascade upstream of the first synaptic relay.
Pro-inflammatory cytokines whose signaling is inhibited include IL-4 (JAK1/JAK3 and JAK1/JAK2), IL-5 (JAK2-STAT3/5, involved in eosinophilia), IL-13 (JAK1/JAK2/TYK2-STAT6, mediating goblet cell hyperplasia and IgE production), and IFN-γ (JAK1/JAK2-STAT1). IL-33, an alarm cytokine released by damaged keratinocytes, signals primarily via the ST2/IL-1RAcP complex and the MyD88/IRAK/NF-κB pathway, activating resident skin mast cells (Di Salvo 2018). Although some experimental data have suggested JAK2/STAT5 phosphorylation in mast cell models, IL-33 signaling is not considered JAK-dependent in the canonical sense, and the impact of JAK inhibition on IL-33-induced mast cell activation remains to be demonstrated.
The pronounced inhibition of JAK2 by ilunocitinib impairs signaling of granulocyte-macrophage colony-stimulating factor (GM-CSF), a JAK2-dependent cytokine involved in the maturation, survival, and activation of dendritic cells and tissue macrophages (Huang 2022). This inhibition of the GM-CSF/JAK2 axis contributes to the anti-inflammatory efficacy of the molecule but justifies monitoring of the myeloid lineage during prolonged treatments. Furthermore, the impact of ilunocitinib on TYK2 blocks signaling mediated by type I interferons (IFN-α/IFN-β), and not only that of IFN-γ (type II). Type I interferons, signaling via the TYK2/JAK1-STAT1/STAT2 pair, constitute the first line of innate antiviral defense. Consequently, inhibition of this pathway by ilunocitinib mechanistically explains the increased susceptibility to viral infections (canine adenovirosis) observed in pharmacovigilance, beyond adaptive immunosuppression alone (Spinelli 2021).
The moderate inhibition of JAK3 by ilunocitinib (IC₅₀ = 16 nM, markedly lower than the inhibitory potency against JAK1 and JAK2) partially preserves adaptive immunity dependent on the common gamma chain (γc). The γc chain is the shared component of IL-2, IL-7, IL-15, and IL-21 receptors, cytokines critical for the proliferation and survival of Natural Killer (NK) cells and memory T lymphocytes (Bonelli 2024). This partial preservation of JAK3/γc signaling distinguishes ilunocitinib from human tofacitinib (a potent JAK3 inhibitor) and contributes to the maintenance of antitumor and antiviral immunosurveillance at the therapeutic dose. The collateral inhibition of IL-12, type I interferons, and IFN-γ, while producing deep lesional clearance, generates a relative immunosuppression inherent to the mechanism of action with regard to host defense cytokines, which must be weighed against the clinical benefit (Huang 2022). Blockade of the IL-4/IL-13/STAT6 pathway could theoretically contribute to reducing total and specific IgE production by B lymphocytes, although this hypothesis has not yet been formally demonstrated in published veterinary clinical studies.
3.4 Pharmacodynamic comparison with other veterinary JAKi
3.4.1 Comparative positioning
Ilunocitinib (Zenrelia), oclacitinib (Apoquel), and atinvicitinib represent three JAK inhibitors with distinct pharmacodynamic profiles. Ilunocitinib primarily inhibits JAK1, JAK2, and TYK2; oclacitinib preferentially targets JAK1 and JAK2 (but with limited functional impact on JAK2-dependent pathways (erythropoietin)); and atinvicitinib is a second-generation selective JAK1 inhibitor (Bonelli 2024). From a dosing standpoint, ilunocitinib offers the advantage of once-daily administration without a loading dose, compared to twice-daily administration for 14 days followed by once-daily dosing for oclacitinib. This dosing difference eliminates the risk of pruritus rebound when transitioning to the maintenance dose, a phenomenon documented with oclacitinib (Olivry 2023, Fukuyama 2017). The inhibition of JAK2 by ilunocitinib, absent from oclacitinib’s profile, may explain the hematological changes observed at high doses but confers a broader anti-inflammatory spectrum covering the IFN-γ pathway (Bonelli 2024).
PART IV — PHARMACOKINETICS
4.1 Absorption
4.1.1 Bioavailability and food effect
Following oral administration, ilunocitinib is rapidly absorbed, with a median time to reach maximum plasma concentration (Tmax) of 1 to 4 hours depending on fasting status (Boerngen 2026). The effect of food on bioavailability has been characterized in detail. Absolute bioavailability is 79.6% (90% CI: 67.6–93.6%) with a meal versus 60.8% (90% CI: 51.6–71.5%) in the fasted state. Mean maximum concentrations (Cmax) at a dose of 0.8 mg/kg are 268 ng/mL (with a meal) versus 122 ng/mL (fasted). Despite this significant food effect under single-dose laboratory conditions, the absence of a clinically significant pharmacokinetic difference was confirmed in the three field studies, in which owners administered the veterinary medicinal product with or without food at their convenience (Forster 2025a, 2025b, 2025c). This discordance between laboratory data and field data is attributed to attenuation of the food effect under repeated dosing conditions.
4.2 Distribution
4.2.1 Distribution parameters
Following intravenous administration, ilunocitinib exhibits a plasma clearance of 0.437 L/h/kg (7.3 mL/min/kg) and a steady-state volume of distribution (Vss) of 1.58 L/kg (Boerngen 2026). A Vss greater than 1 L/kg indicates extensive tissue distribution, exceeding the plasma compartment and total body water volume. Tissue distribution studies revealed high accumulation in bile, liver, and kidneys, organs involved in the metabolism and elimination of the molecule. Penetration across the blood-brain barrier is low, consistent with the absence of neurological effects observed in the 6-month safety study (Kuntz 2025).
4.3 Metabolism and elimination
4.3.1 Elimination pathways and half-life
Elimination of ilunocitinib follows a dual pathway: approximately 50% of the administered dose is eliminated via the biliary and fecal route, and approximately 30% via the urinary route, representing a recovery rate of approximately 80% at 48 hours (Boerngen 2026). The terminal elimination half-life is 4.4 hours after intravenous administration and approximately 5 hours after oral administration. This relatively short half-life is compatible with once-daily administration and explains the absence of clinically significant accumulation observed with repeated administration: the mean accumulation ratio over AUC (area under the curve) with multiple-dose administration is approximately 1.26 (Boerngen 2026). The 6-month safety study confirmed AUC accumulation ratios of 1.2 to 1.6 depending on dose groups, with no difference between males and females (Kuntz 2025).
4.4 Dose proportionality
4.4.1 Linearity of exposure
The increase in systemic exposure (AUC and Cmax) is linear but not proportional to dose over the dosing interval of 0.4 to 4.0 mg/kg, with a statistically significant intercept in regression analysis (Boerngen 2026). This phenomenon is attributed to solubility-limited absorption at high intraluminal concentrations, consistent with the physicochemical properties of the molecule (low aqueous solubility). This non-proportionality was confirmed in the multiple-dose safety study (Kuntz 2025), where exposure at 4.0 mg/kg (5X) was less than five times the exposure at the therapeutic dose.
4.5 Pharmacokinetics under real clinical conditions
4.5.1 Field data
Pharmacokinetic data collected in the three field clinical trials (placebo-controlled studies and comparative study with oclacitinib) confirm the absence of a clinically significant difference related to feeding status under real use conditions (Forster 2025a, 2025b, 2025c). The consistent efficacy observed regardless of administration with or without food represents a practical advantage for therapeutic compliance by owners, particularly during prolonged treatments in atopic dogs.
PART V — INDICATIONS AND DOSAGE
5.1 Approved indications
5.1.1 Regulatory framework
Zenrelia (ilunocitinib tablets) is indicated for the control of pruritus associated with allergic dermatitis and the control of atopic dermatitis in dogs aged at least 12 months. The indication covers pruritus associated with allergic dermatitis broadly, including atopic dermatitis (sensitization to aeroallergens), flea allergy dermatitis (FAD), and food hypersensitivity with a pruritogenic component, making it a versatile treatment for canine allergic dermatitis. The distinction between CAD and allergic dermatitis has been documented by separate clinical trials (Forster 2025a for allergic dermatitis, Forster 2025b for CAD).
5.2 Dosage and route of administration
5.2.1 Dosing regimen
The recommended therapeutic dose is 0.6 to 0.8 mg/kg of ilunocitinib as a single oral administration once daily, with or without food. The individual dose is calculated by combining the four available tablet strengths (4.8, 6.4, 8.5, and 15 mg) based on the animal’s body weight. No loading dose is required: treatment starts directly at the definitive daily dose, which simplifies the dosing regimen compared to oclacitinib. No maximum treatment duration is specified in the product label, given the absence of clinically significant accumulation and the tolerability documented over 112 days in clinical trials and 182 days in the safety study.
5.3 Onset and duration of action
5.3.1 Kinetics of therapeutic response
The antipruritic efficacy of ilunocitinib is detectable within the first 24 hours and becomes statistically significant as early as day 3: in the allergic dermatitis study, 29% of treated dogs showed a reduction in pruritus of 50% or more (PVAS) by the third day of treatment (Forster 2025a). By day 7, the therapeutic success rate (≥50% reduction in PVAS) was 25.4% in the ilunocitinib group versus 7.7% in the placebo group (p = 0.006). Efficacy on skin lesions, assessed by the CADESI-04 score (Olivry 2014), becomes significant as early as day 14. At day 28, 83% of treated dogs achieved the therapeutic success criterion versus 31% under placebo (p < 0.001) in the European pivotal study on CAD (Forster 2025b). Clinical remission, defined as a PVAS score below 2 (corresponding to a “normal” level of pruritus) or a CADESI-04 score below 10, was achieved in two-thirds of dogs after 4 months of treatment (Forster 2025b).
PART VI — CLINICAL EFFICACY STUDIES
6.1 Pivotal placebo-controlled study (Europe/USA)
6.1.1 Protocol and results
The pivotal trial published by Forster, Trout, Despa et al. in Veterinary Dermatology (2025) evaluated the efficacy and safety of ilunocitinib in the control of canine atopic dermatitis. The protocol was a randomized, double-masked, placebo-controlled trial conducted in 25 veterinary clinics in Great Britain and Germany. Two hundred sixty-eight dogs with confirmed CAD were enrolled: 181 in the ilunocitinib group (0.6–0.8 mg/kg/day) and 87 in the placebo group, for a duration of 112 days (Forster 2025b). The most represented breeds were the Labrador Retriever, French Bulldog, West Highland White Terrier, and Golden Retriever (5.6%). The age of enrolled dogs ranged from 1.0 to 17.3 years.
The primary endpoint was the therapeutic success rate at day 28, defined as a reduction of 50% or more in the PVAS score (pruritus assessed by the owner) or the CADESI-04 score (skin lesions assessed by the investigator). Results demonstrated statistically significant superiority of ilunocitinib over placebo at all evaluation time points. At day 28, 83% of dogs in the ilunocitinib group achieved the success criterion on PVAS versus 31% in the placebo group (p < 0.001) (Forster 2025b). The proportion of dogs achieving clinical remission of pruritus (PVAS < 2) was significantly higher in the ilunocitinib group as early as day 7 (p < 0.05), and remission of skin lesions (CADESI-04 < 10) as early as day 14 (p < 0.05). No dog in the placebo group had reached the CADESI-04 < 10 threshold by day 28. The cumulative proportion of dogs withdrawn from the study for perceived lack of efficacy was significantly higher in the placebo group. The 112-day treatment was well tolerated.
6.2 Comparative study: ilunocitinib vs. oclacitinib
6.2.1 Comparative results
The comparative trial published by Forster, Boegel, Despa et al. in Veterinary Dermatology (2025) evaluated the efficacy and safety of ilunocitinib in direct comparison with oclacitinib in dogs with CAD. The trial was randomized, masked, and conducted in 25 centers across four European countries. Three hundred thirty-eight dogs were randomized to receive either oclacitinib (0.4–0.6 mg/kg twice daily for 14 days, then once daily) or ilunocitinib (0.6–0.8 mg/kg once daily) for a maximum of 112 days (Forster 2025c). The reduction in pruritus (PVAS) and CADESI-04 scores was comparable between the two groups during the first 14 days of treatment (days 0–14), corresponding to the twice-daily phase of oclacitinib. However, between day 14 and day 28, PVAS scores increased in the oclacitinib group (rebound phenomenon upon transition to maintenance dose) while continuing to decrease in the ilunocitinib group. From day 28 to day 112, mean PVAS and CADESI-04 scores were significantly lower in the ilunocitinib group (p ≤ 0.003 and p ≤ 0.023, respectively). A significantly higher number of dogs treated with ilunocitinib achieved clinical remission of pruritus (PVAS < 2) from day 28 to day 112. The investigators’ overall subjective assessment of therapeutic response was significantly better for ilunocitinib from day 28 to day 112 (p ≤ 0.002). Tolerability profiles were comparable between the two groups (Forster 2025c).
6.3 Studies on allergic dermatitis (non-atopic)
6.3.1 US placebo-controlled trial
The randomized, double-masked, placebo-controlled trial published by Forster, Trout, Despa et al. in Veterinary Dermatology (2025) evaluated ilunocitinib in 306 dogs presenting with severe pruritus and a presumptive diagnosis of allergic dermatitis, enrolled in 15 veterinary clinics in the United States (Forster 2025a). Dogs were randomized to receive ilunocitinib (n = 206; 0.6–0.8 mg/kg) or placebo (n = 100) once daily for 28 days, with a safety follow-up of 112 days. More than 90% of dogs in each group had a presumptive diagnosis of CAD, with concomitant diagnoses including contact dermatitis (23.3%), FAD (15.5%), and food hypersensitivity (24.3%). The primary endpoint was the therapeutic success rate at day 7, defined as a ≥50% reduction in PVAS for at least 5 of the first 7 days. At day 7, 25.4% of dogs treated with ilunocitinib achieved this criterion versus 7.7% under placebo (p = 0.006). As early as day 3, the proportion of dogs showing a ≥50% reduction in PVAS was significantly higher in the ilunocitinib group (p < 0.01). At day 28, 51.8% of treated dogs achieved clinical remission (PVAS < 2) versus 12.7% under placebo (p < 0.05). Manifestations of dermatitis improved as early as day 7. The 112-day treatment was well tolerated (Forster 2025a).
PART VII — SAFETY DATA
7.1 Target species safety study (6 months)
7.1.1 Protocol
The safety study published by Kuntz, Gabor, and Toutain in BMC Veterinary Research (2025) constitutes the cornerstone of the ilunocitinib safety dossier. The protocol, conducted in accordance with VICH GL43 recommendations, was a randomized, masked, parallel-group trial including 40 healthy Beagles (20 males and 20 females), aged 11 to 12 months, with a mean body weight of 9.85 to 10.46 kg (Kuntz 2025). The animals were assigned to one untreated control group and four groups receiving ilunocitinib at daily doses of 0.8 mg/kg (1X, maximum recommended therapeutic dose), 1.6 mg/kg (2X), 2.4 mg/kg (3X), and 4.0 mg/kg (5X) for 182 days. All animals were fed within 30 minutes prior to administration. Safety assessments included general clinical observations, complete physical and neurological examinations (every two weeks), ophthalmic examinations, hematology, serum biochemistry, coagulation, peripheral blood lymphocyte immunophenotyping, body weight, food consumption, pharmacokinetic sampling, and macroscopic and histopathological organ examination at study end.
7.1.2 Results
At the therapeutic dose (1X = 0.8 mg/kg), no clinically significant adverse effects were reported. No treatment effect was observed on body weight, food consumption, physical and neurological examinations, urinalysis, peripheral blood lymphocyte immunophenotype (total CD5+ T cells, CD4+ Th, CD8+ Tc, CD21+ B cells, monocytes), or ophthalmic examinations (Kuntz 2025). At higher doses, interdigital skin lesions (furunculosis, interdigital follicular cysts) appeared in a dose-dependent manner, more frequent at doses ≥3X, resolving spontaneously or following standard antimicrobial treatment. A slight decrease in erythrocytic parameters (hemoglobin, hematocrit, red blood cell count, MCHC, MCH) was observed at doses ≥3X, with values remaining within reference limits at the 1X level. A decrease in eosinophils was noted in females at doses ≥3X (p = 0.0050). An increase in fibrinogen and C-reactive protein at doses ≥3X (p < 0.0001 for fibrinogen) was interpreted as an acute phase response secondary to interdigital skin lesions. Cutaneous papillomas were observed in 2 dogs at the 5X dose, with expected spontaneous resolution. A slight modification of adrenal gland weights was noted without histopathological correlation. No hepatic, renal, ocular, or neurological effects were documented. No mortality or serious adverse events occurred during the 182-day study, including at 5X the maximum recommended dose, and all treated animals completed the intended study duration (Kuntz 2025).
7.2 Safety in field clinical trials
7.2.1 Compilation of tolerability data
Tolerability data compiled from the three clinical trials (Forster 2025a, 2025b, 2025c) cover more than 500 dogs treated with ilunocitinib under real-world conditions. The most frequently reported adverse effects were gastrointestinal disorders and lethargy. The FDA registration dossier specifies incidences in the field trials: diarrhea in 12.2% and urinary tract infections in 6.3% (FDA 2024). The overall incidence of gastrointestinal adverse effects in the allergic dermatitis study (Forster 2025a) was comparable between the treated group and the placebo group at day 28: 47.6% versus 49.0%, demonstrating that ilunocitinib does not increase the incidence of digestive disorders compared to placebo. Neoplastic conditions (benign and malignant) were observed during the clinical studies, motivating the recommendation for monitoring in dogs with a history of recurrent neoplasia. The pivotal CAD study reported a slight increase in albumin in the ilunocitinib group at days 56, 84, and 112, without determined clinical significance (Forster 2025b). An isolated case of cyclic neutropenia was identified in one treated dog, considered unrelated to treatment. Platelet counts remained within reference limits across all studies (Forster 2025b, Kuntz 2025). In the comparative study with oclacitinib, adverse event rates were similar between the two groups, confirming a comparable tolerability profile (Forster 2025c).
7.3 Post-marketing data
7.3.1 International pharmacovigilance
Since the commercial launch in Brazil (first market), then in the United States, Japan, Canada, and Europe, more than 800,000 dogs have been treated with Zenrelia as of September 2025, according to data communicated by Elanco. Post-marketing pharmacovigilance has led to the identification of signals concerning susceptibility to infections (adenoviral hepatitis, pancreatitis) and a risk of inadequate vaccine immune response, integrated into the label warnings. In September 2025, retrospective PCR analysis of hepatic tissues from the two euthanized dogs in the initial vaccine response study revealed the presence of CAV-1 (canine adenovirus type 1, the causative agent of infectious canine hepatitis through natural infection), suggesting that a natural epizootic, and not vaccine-induced disease, was the cause of the clinical deterioration in these animals (Fent 2025a). The FDA concluded that the totality of evidence supported removal of the risk of fatal vaccine-induced disease from modified live virus vaccines from the labeling.
PART VIII — CONTRAINDICATIONS AND PRECAUTIONS FOR USE
8.1 Absolute contraindications
8.1.1 Exclusion situations
Ilunocitinib must not be used in dogs under 12 months of age, in dogs with severe infections, or in dogs with confirmed malignant neoplasia, demodicosis, or an immunosuppressed state (hyperadrenocorticism, for example), situations in which the molecule has not been evaluated. The absence of data in breeding dogs, pregnant or lactating females constitutes a relative contraindication. Access to treatment is therefore reserved for adult dogs in good general health and up to date on their vaccinations.
8.2 Vaccination warning
8.2.1 Vaccine response studies
The effect of ilunocitinib administration on vaccination against canine parvovirus (CPV), canine distemper virus (CDV), canine adenovirus-2 (CAV-2), canine parainfluenza virus (CPiV), and inactivated rabies vaccine (RV) was studied in two separate protocols. The primary vaccination study (Fent 2025a) was conducted in 10-month-old seronegative Beagles, vaccination-naive, receiving 2.4 mg/kg (3X the maximum recommended dose) for 89 days. During this study, a coccidiosis epizootic (Cystoisospora canis) and a confirmed case of infectious canine hepatitis (CAV-1) occurred, confounding the interpretation of results. Two treated dogs were euthanized due to deterioration of their general condition. Based on assessment of serological antibody titers, an adequate immune response to common canine modified live vaccines (CAV-2, CDV, and CPV) was observed following primary vaccination at day 28. The response to primary vaccination with CPiV (non-core vaccine) in treated animals was 4 out of 6 above the threshold compared to 6 out of 8 above the threshold for control animals. A delayed or reduced response to RV was observed (Fent 2025a).
The booster vaccination study (Fent 2025b), published in BMC Veterinary Research, evaluated the serological response to booster vaccination in 10-month-old dogs previously vaccinated according to standard recommendations, receiving ilunocitinib at 1X or 3X the recommended dose (0.6–0.8 or 1.8–2.4 mg/kg, respectively) for 56 days. Results demonstrated that treatment with ilunocitinib at 1X and 3X doses did not significantly attenuate the serological response to CAV-2, CPV, CDV, or rabies booster vaccinations compared to untreated control animals (Fent 2025b). All treated dogs had antibody titers at or above the protective threshold for rabies, CAV-2, and CPV at days 43 and 56 post-vaccination. The current label maintains the recommendation to discontinue Zenrelia at least 28 days to 3 months before any vaccination and not to resume treatment until at least 28 days after vaccination. Dogs must be up to date on their vaccinations before initiating treatment.
The FDA’s conclusion is that the therapeutic dose (0.6–0.8 mg/kg) presents no documented risk of fatal vaccine-induced disease, following label revision in September 2025.
8.3 Infectious risks and monitoring
8.3.1 Infectious susceptibility and follow-up protocol
The immunomodulation induced by JAK kinase inhibition theoretically increases susceptibility to opportunistic infections. Infections documented in the atopic dermatitis clinical studies and in post-marketing pharmacovigilance include bacterial skin infections (pyoderma), respiratory tract infections, urinary tract infections, reactivation of Demodex canis, and intestinal parasitic infections (coccidiosis). Canine adenovirosis (hepatitis and pancreatitis) has been identified as a pharmacovigilance signal. A clinical monitoring protocol is recommended, including regular physical examination, a control complete blood count (looking for abnormalities in erythrocytic parameters: hemoglobin, hematocrit, platelet count, and red blood cell count), and increased vigilance regarding cutaneous, respiratory, and digestive infections (Kuntz 2025).
8.4 Neoplastic risk
8.4.1 Benefit/risk balance
Benign and malignant neoplastic conditions were observed during ilunocitinib clinical studies, without a causal link having been formally established (Forster 2025b). By analogy with the accumulated data in human medicine on JAKi (tofacitinib, baricitinib, upadacitinib), monitoring for the emergence of neoplasias during prolonged use is recommended, particularly in dogs with a history of recurrent neoplasia. The benefit/risk balance must be evaluated individually for each patient.
8.5 Populations at particular risk
8.5.1 Unevaluated populations
Elderly dogs, breeds with particular infectious susceptibility (brachycephalic breeds, breeds predisposed to demodicosis), and dogs receiving concomitant immunosuppressive therapy constitute higher-risk populations requiring enhanced monitoring. The absence of data in dogs under 12 months of age, pregnant or lactating females, and breeding dogs requires careful extrapolation. Combination with other immunosuppressants (glucocorticoids, cyclosporine) was not evaluated within the clinical development program (Kuntz 2025).
PART IX — DRUG INTERACTIONS
9.1 Pharmacodynamic interactions
Combination of ilunocitinib with other immunosuppressive agents (systemic glucocorticoids, cyclosporine, azathioprine, mycophenolate mofetil) may potentiate immunosuppression and increase infectious risk (EMA 2025). This combination is not recommended without a rigorous assessment of the benefit/risk balance on a case-by-case basis. Data from clinical trials were collected under conditions where anti-infective treatments (antibiotics, antifungals, antiparasitics) and non-steroidal topicals were permitted concomitantly, and vaccinations were allowed according to study protocol (Forster 2025a, 2025b, 2025c). The combination of ilunocitinib and allergen-specific immunotherapy (ASIT, desensitization) constitutes a clinical perspective supported by available data: Fennis et al. (2022) demonstrated in a prospective trial of 48 atopic dogs that concurrent administration of oclacitinib and ASIT did not impair the lymphocyte modulation induced by immunotherapy, with clinical response rates remaining comparable to those of dogs receiving ASIT alone. By pharmacodynamic extrapolation (shared JAK mechanism), maintenance of ilunocitinib during desensitization induction phases appears compatible with ASIT efficacy, as desensitization acts via induction of tolerance by regulatory T lymphocytes and blocking IgG — immunological mechanisms distinct from the JAK-STAT pathway (Fennis 2022).
9.2 Pharmacokinetic interactions
9.2.1 P-glycoprotein substrate
Ilunocitinib is a potential substrate of P-gp (ABCB1) transporters at the intestinal level, by analogy with baricitinib and tofacitinib (Spinelli 2021). Drugs inhibiting P-gp (ketoconazole, itraconazole, cyclosporine, verapamil) could theoretically increase the bioavailability of ilunocitinib by reducing intestinal efflux, while P-gp inducers (rifampicin, phenobarbital) could decrease it (Payne 2015). Their co-administration with ilunocitinib could increase systemic exposure of the latter through competition on the ABCB1 efflux transporter.
9.2.2 Interactions with canine cytochrome P450
The influence of hepatic cytochrome P450 on the metabolism of ilunocitinib has not been fully characterized in vivo in dogs, although metabolic data suggest a significant contribution of the biliary pathway to elimination (Boerngen 2026). Canine CYP2D15, the ortholog of human CYP2D6, metabolizes many psychotropic and cardiovascular drugs. Clomipramine (CLOMICALM tablets 5–20–80 mg, Elanco), a tricyclic frequently prescribed for treatment of anxiety disorders in atopic dogs (separation anxiety, lick granuloma), is a documented inhibitor of canine CYP2D15 (Aidasani 2008). Concurrent administration of ilunocitinib and clomipramine could theoretically modify the metabolic profile of either compound if CYP2D15 contributes significantly to their biotransformation. Caution is warranted when concurrently administering drugs that are substrates or inhibitors of efflux transporters or hepatic CYPs (EMA 2025).
PART X — IN-DEPTH IMMUNOLOGICAL DATA
10.1 Impact on lymphocyte subpopulations
Peripheral blood lymphocyte immunophenotyping performed in the 6-month safety study (Kuntz 2025) demonstrated the absence of a significant effect of ilunocitinib on peripheral lymphocyte subpopulations CD5+ total T cells, CD4+ helper T cells, CD8+ cytotoxic T cells, CD21+ B cells, and monocytes at the therapeutic dose (0.8 mg/kg). This preservation of the circulating immunophenotype at the clinical dose contrasts with human tofacitinib data, for which dose-dependent reductions in NK (natural killer) lymphocytes and CD8+ T lymphocytes have been documented in human rheumatology (Strober 2013). The absence of lymphocyte population disruption at the therapeutic dose constitutes a significant argument for immunological tolerability, suggesting that maintenance of adaptive immunity is compatible with the antipruritic and anti-inflammatory efficacy of the molecule. However, regular hematological monitoring remains recommended during prolonged use, as very long-term safety data (beyond 6 months) are not yet available from controlled studies.
10.2 Impact on erythropoiesis
A slight, dose-dependent decrease in erythrocytic parameters (hemoglobin, hematocrit, red blood cell count, MCHC, MCH) was documented at doses ≥3X in the safety study (Kuntz 2025). This phenomenon is attributed to inhibition of JAK2/EPO (erythropoietin) signaling, a critical pathway of medullary erythropoiesis. Binding of EPO to its homodimeric receptor activates the JAK2/JAK2 pair, inducing STAT5 phosphorylation and transcription of survival and proliferation genes in erythroid progenitors. Inhibition of JAK2 by ilunocitinib leads to a dose-dependent attenuation of this cascade, with clinical consequences remaining subliminal at the therapeutic dose (values remaining within reference limits), but potentially becoming significant in cases of prolonged overdose. This observation is consistent with human JAKi data: baricitinib induces a moderate decrease in hemoglobin in 2 to 5% of human patients treated for rheumatoid arthritis, and tofacitinib has been associated with cases of anemia requiring dose reduction (Strober 2013). Monitoring of the complete blood count, particularly hematocrit and hemoglobin, is recommended during long-term use of ilunocitinib, with cautious dose advancement in anemic dogs or those with compromised medullary health.
10.3 Effect on the Th2 pathway and the IL-4/IL-13 axis: therapeutic implications beyond pruritus
Inhibition of IL-4/IL-13 signaling through blockade of the JAK1/JAK3 and JAK1/JAK2/TYK2 pairs constitutes one of the central pharmacodynamic mechanisms of ilunocitinib, with potentially broader therapeutic implications than mere pruritus control. IL-4, binding to its type I receptor (IL-4Rα/γc, JAK1/JAK3) or type II receptor (IL-4Rα/IL-13Rα1, JAK1/TYK2), activates STAT6 phosphorylation, a key transcription factor for Th2 polarization. Phosphorylated STAT6 induces transcription of genes encoding the immunoglobulin ε chain (isotype class switching toward IgE), the chemokine receptors CCR4 and CCR8, and Th2 pro-inflammatory mediators (IL-5, IL-13) (Olivry 2010). Inhibition of this pathway by ilunocitinib could theoretically reduce total and specific IgE production, contribute to epidermal barrier restoration (IL-13 impairs expression of keratinocyte junction proteins), and exert an anti-inflammatory effect beyond the scope of symptomatic control of pruritus and itching. These mechanistic hypotheses constitute future research directions, as current clinical data do not document a reduction in serum IgE levels under ilunocitinib in published trials. The comparative study with oclacitinib (Forster 2025c) revealed a more pronounced and sustained decrease in skin lesions (CADESI-04) under ilunocitinib from day 28 to day 112, suggesting a cutaneous anti-inflammatory effect going beyond simple antipruritic control and possibly related to the additional inhibition of TYK2 (IL-12/IL-23-Th1/Th17 pathway).
Research perspectives
The arrival of ilunocitinib in the veterinary dermatology therapeutic arsenal opens several investigational avenues. Very long-term safety data (beyond 12 months) under real clinical conditions remain necessary, particularly to characterize cumulative neoplastic and infectious risk. In human medicine, the ORAL Surveillance study highlighted an increased risk of major cardiovascular events and neoplasias under tofacitinib compared to anti-TNF agents in rheumatological patients over 50 years of age, leading the FDA to issue a boxed warning for all human JAKi. Extrapolation of these data to veterinary medicine remains speculative, as canine cardiovascular pathophysiology and patient life expectancy are fundamentally different, but oncological and infectious vigilance is warranted during prolonged treatments.
The impact of ilunocitinib on the cutaneous microbiome of atopic dogs, on epidermal barrier restoration, and on serum biomarkers (total and specific IgE, circulating cytokines) merits prospective evaluation. Transcriptomic studies conducted on skin biopsies from atopic dogs treated with oclacitinib have demonstrated partial normalization of intercellular junction protein expression and antimicrobial peptides (Marsella 2021), suggesting that JAK inhibition may exert a barrier restoration effect beyond mere symptomatic control. Similar studies with ilunocitinib, leveraging its complementary inhibition profile (TYK2), would allow documentation of a potential therapeutic advantage on the biology of the skin barrier.
The combination of ilunocitinib and allergen-specific immunotherapy (ASIT) could constitute a combined strategy associating rapid symptomatic control with long-term modification of the allergic immune response. ASIT, whose efficacy is documented in 50 to 75% of atopic dogs, depending on studies and protocols, after 6 to 12 months of protocol (Olivry 2015), acts by induction of regulatory T lymphocytes producing IL-10 and TGF-β, and by isotypic switching of IgE toward blocking IgG4. The maintenance of regulatory T lymphocyte populations under ilunocitinib, suggested by the absence of CD4+ immunophenotype disruption in the Kuntz study (2025), is an argument in favor of the compatibility of this combination. Combined trials are awaited to document this hypothesis.
Comparative data with lokivetmab and second-generation JAK inhibitors currently under veterinary development will allow positioning of ilunocitinib within the therapeutic algorithm. Evaluation of anti-IL-4Rα canine biotherapies (analogous to human dupilumab) constitutes an exploratory research direction. Characterization of CAD susceptibility genes, awaited for two decades, could ultimately allow identification of predictive biomarkers of therapeutic response to JAKi. Population pharmacokinetic studies, integrating breed, age, weight, and hepatic function covariates, constitute an indispensable complement to current data for optimizing individualized dosing regimens.
Ilunocitinib thus emerges as a significant therapeutic advance in the management of canine atopic dermatitis and associated allergic dermatitis, offering a favorable tolerability profile at the therapeutic dose, efficacy documented by high-level evidence controlled trials, and a simplified once-daily dosing regimen. Access to international pharmacovigilance data on a growing cohort of treated dogs — more than 800,000 as of September 2025, with commercial deployment in Europe, North America, Brazil, and Japan — will allow further characterization of rare clinical manifestations and drug interactions under real-world conditions. The state of knowledge in 2025–2026 positions this molecule as a first-line tool for the dermatologist practitioner and general practitioner faced with canine allergic dermatitis, whose integration into a multimodal therapeutic strategy — combining environmental allergen control, epidermal barrier restoration with emollient topicals, management of bacterial and fungal superinfections, and where applicable allergen-specific immunotherapy — remains the key to optimal and durable management of CAD. The positioning of Zenrelia relative to existing molecules (oclacitinib, lokivetmab, cyclosporine) and molecules under development (atinvicitinib, second-generation selective inhibitors) will become clearer as comparative publications and large-scale clinical experience accumulate.
References
Aidasani D, Zaya MJ, Muppal JG, Locuson CW. In vitro drug-drug interaction screening of canine cytochrome P450 enzymes with clomipramine and related tricyclics. Drug Metab Dispos. 2008;36:1512-1518.
Boerngen K, Patel Y, Pittorino M, Toutain CE. Pharmacokinetics of ilunocitinib, a new Janus kinase inhibitor, in dogs. J Vet Pharmacol Ther. 2026;49(1):7–16.
Bonelli M, Kerschbaumer A, Kastrati K, Ghoreschi K, Gadina M, Heinz LX, et al. Selectivity, efficacy and safety of JAKinibs: new evidence for a still evolving story. Ann Rheum Dis. 2024;83:139-160.
Chaudhary SK, Singh SK, Kumari P, Kanwal S, Soman SP, Choudhury S, et al. Alterations in circulating concentrations of IL-17, IL-31 and total IgE in dogs with atopic dermatitis. Vet Dermatol. 2019;30:383-e114.
Choy EH. Clinical significance of Janus kinase inhibitor selectivity. Rheumatology. 2019;58:953-962.
Clark JD, Flanagan ME, Telliez JB. Discovery and development of Janus kinase (JAK) inhibitors for inflammatory diseases. J Med Chem. 2014;57:5023-5038.
Cosgrove SB, Wren JA, Cleaver DM, Walsh KF, Follis SI, King VI, et al. Long-term compassionate use of oclacitinib in dogs with atopic and allergic skin disease: safety, efficacy and quality of life. Vet Dermatol. 2015;26:171-179.
Datsi A, Steinhoff M, Ahmad F, Alam M, Buddenkotte J. Interleukin-31: the “itchy” cytokine in inflammation and therapy. Allergy. 2021;76:2982-2997.
Di Salvo E, Ventura-Spagnolo E, Casciaro M, Navarra M, Gangemi S. IL-33/IL-31 axis: a potential inflammatory pathway. Mediators Inflamm. 2018;2018:3858032.
Drechsler Y, Dong C, Clark DE, Kaur G. Canine atopic dermatitis: prevalence, impact, and management strategies. Vet Med (Auckl). 2024;15:15-29.
EMA. European Public Assessment Report (EPAR) Zenrelia™ (ilunocitinib). European Medicines Agency; 2025.
Favrot C, Fischer N, Olivry T, Zwickl L, Audergon S, Rostaher A. Atopic dermatitis in West Highland White Terriers — Part I: Natural history of atopic dermatitis in the first three years of life. Vet Dermatol. 2020;31:106-110.
FDA. New Animal Drug Application NADA 141-585 Zenrelia™ (ilunocitinib tablets). US Food and Drug Administration; 2024.
Fennis M, Vroom M, Broeckx BJG, Vandenabeele S, De Cock H. Concurrent administration of oclacitinib and allergen-specific immunotherapy in dogs with atopic dermatitis. Vet Dermatol. 2022;33:344-e85.
Fent GM, Despa S, Gabor L, Earll M, McCandless EE, O’Kelley S, et al. Response to primary canine core vaccination in 10-month-old seronegative dogs treated with three times the recommended therapeutic dose of ilunocitinib tablets (Zenrelia™). BMC Vet Res. 2025a;21:461.
Fent GM, Jacela J, Plazola-Ortiz R, Olps J, McCandless EE, Toutain CE, et al. Immunologic response to first booster vaccination in dogs treated with Zenrelia™ (ilunocitinib tablets) at up to three times the recommended therapeutic dose compared to untreated controls. BMC Vet Res. 2025b;21:481.
Forster S, Trout CM, Despa S, Boegel A, Berger D, King S. Efficacy and field safety of ilunocitinib for the control of allergic dermatitis in client-owned dogs: a multicenter, double-masked, randomised, placebo-controlled clinical trial. Vet Dermatol. 2025a;0:1-11.
Forster S, Trout CM, Despa S, Boegel A, Berger D, King S. Efficacy and field safety of ilunocitinib for the control of atopic dermatitis in client-owned dogs: a multicentre, double-masked, randomised, placebo-controlled clinical trial. Vet Dermatol. 2025b;0:1-13.
Forster S, Boegel A, Despa S, Trout C, King S. Comparative efficacy and safety of ilunocitinib and oclacitinib for the control of pruritus and associated skin lesions in dogs with atopic dermatitis. Vet Dermatol. 2025c;36:165-176.
Fukuyama T, Ganchingco JR, Bäumer W. Demonstration of rebound phenomenon following abrupt withdrawal of the JAK1 inhibitor oclacitinib. Eur J Pharmacol. 2017;794:20-26.
Gonzales AJ, Humphrey WR, Messamore JE, Fleck TJ, Fici GJ, Shelly JA, et al. Interleukin-31: its role in canine pruritus and naturally occurring canine atopic dermatitis. Vet Dermatol. 2013;24:48-53.
Gonzales AJ, Bowman JW, Fici GJ, Zhang M, Mann DW, Mitton-Fry M. Oclacitinib (APOQUEL) is a novel Janus kinase inhibitor with activity against cytokines involved in allergy. J Vet Pharmacol Ther. 2014;37:317-324.
Hensel P, Santoro D, Favrot C, Hill P, Griffin C. Canine atopic dermatitis: detailed guidelines for diagnosis and allergen identification. BMC Vet Res. 2015;11:196.
Hillier A, Griffin CE. The ACVD task force on canine atopic dermatitis (I): incidence and prevalence. Vet Immunol Immunopathol. 2001;81:147-151.
Huang JT, Doering KA, Fang X. Mechanism of action underlying the efficacy of JAK inhibitors in skin diseases. J Drugs Dermatol. 2022;21:234-241.
Kuntz EA, Gabor L, Toutain CE. Safety of ilunocitinib tablets (Zenrelia™) after once daily oral administration in dogs. BMC Vet Res. 2025;21:144.
Marsella R. Advances in our understanding of canine atopic dermatitis. Vet Dermatol. 2021;32:547-e151.
Michels GM, Ramsey DS, Walsh KF, Martinon OM, Mahabir SP, Hoevers JD, et al. A blinded, randomized, placebo-controlled, dose determination trial of lokivetmab (ZTS-00103289), a caninized, anti-canine IL-31 monoclonal antibody in client owned dogs with atopic dermatitis. Vet Dermatol. 2016;27:478-e129.
Nemmer JM, Stander S. New developments in the pathophysiology of pruritus. Dermatol Ther (Heidelb). 2021;11:1795-1815.
Olivry T, Bizikova P. A systematic review of the evidence of reduced allergenicity and clinical benefit of food hydrolysates in dogs with cutaneous adverse food reactions. Vet Dermatol. 2010;21:32-41.
Olivry T, Saridomichelakis M, Nuttall T, Bensignor E, Griffin CE, Hill PB, et al. Validation of the canine atopic dermatitis extent and severity index (CADESI)-4, a simplified severity scale for assessing skin lesions of atopic dermatitis in dogs. Vet Dermatol. 2014;25:77-85.
Olivry T, DeBoer DJ, Favrot C, Jackson HA, Mueller RS, Nuttall T, et al. Treatment of canine atopic dermatitis: 2015 updated guidelines from the International Committee on Allergic Diseases of Animals (ICADA). BMC Vet Res. 2015;11:210.
Olivry T, Lokianskiene V, Blanco A, et al. A randomised controlled trial testing the rebound-preventing benefit of four days of prednisolone during the induction of oclacitinib therapy in dogs with atopic dermatitis. Vet Dermatol. 2023;34:99-106.
O’Shea JJ, Schwartz DM, Villarino AV, Gadina M, McInnes IB, Laurence A. The JAK-STAT pathway: impact on human disease and therapeutic intervention. Annu Rev Med. 2015;66:311-328.
Payne JT, Danielson JR, Bennett BS, Wheeler J, Krebber R. Interaction of transporter proteins with P-glycoprotein inhibitors and substrates in vitro. Drug Metab Dispos. 2015;43:1199-1207.
Pucheu-Haston CM, Bizikova P, Marsella R, Santoro D, Nuttall T, Eisenschenk MN. Review: Genetics, epigenetics and allergy of canine atopic dermatitis. Vet Dermatol. 2015;26:84-e26.
Santoro D, Marsella R, Pucheu-Haston CM, Eisenschenk MN, Nuttall T, Bizikova P. Review: Pathogenesis of canine atopic dermatitis: skin barrier and host-micro-organism interaction. Vet Dermatol. 2015;26:84-e25.
Spinelli FR, Meylan F, O’Shea JJ, et al. JAK inhibitors: ten years after. Eur J Immunol. 2021;51:1615-1627.
Steffan J, Favrot C, Mueller R. A systematic review and meta-analysis of the efficacy and safety of cyclosporin for the treatment of atopic dermatitis in dogs. Vet Dermatol. 2006;17:3-16.
Strober B, Buonanno M, Clark JD, et al. Effect of tofacitinib, a Janus kinase inhibitor, on haematological parameters during 12 weeks of psoriasis treatment. Br J Dermatol. 2013;169:992-999.
Virtanen A, Palmroth M, Liukkonen S, et al. Differences in JAK isoform selectivity among different types of JAK inhibitors evaluated for rheumatic diseases through in vitro profiling. Arthritis Rheumatol. 2023;75:2054-2061.